THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
A widely used technique for identifying subtypes of neurons in the brain may be flawed, and may have provided incorrect tallies of a key neuron type in autism. These new findings call into question some of the evidence supporting a popular theory of autism, and may have implications for other fields1.
In autism research, the method has been used to track a class of inhibitory neurons, or interneurons, that dampen signaling activity in the brain. This class of interneurons produces a protein called parvalbumin.
Studies in animal models and human brains have suggested that people with autism have a dearth of parvalbumin interneurons. Those reports supported the theory that autism stems from an imbalance between excitation and inhibition in the brain, leading to an overactive brain.
However, the new study found that certain mouse models of autism have about the same number of these interneurons as controls do, but those cells produce less parvalbumin.
The findings may call for a revisit of the earlier research — that is, those studies might have mistakenly interpreted the abnormally low levels of parvalbumin as absence of the neurons themselves.
“If all the groups that reported loss of parvalbumin interneurons went back and certified whether [their findings] are really due to a loss of neurons or whether they’re really due to a down-regulation of parvalbumin, this could completely change the interpretation of these results,” says lead researcher Beat Schwaller, associate professor of anatomy at the University of Fribourg in Switzerland. Schwaller’s study appeared 27 January in Molecular Brain.
Marker clues:
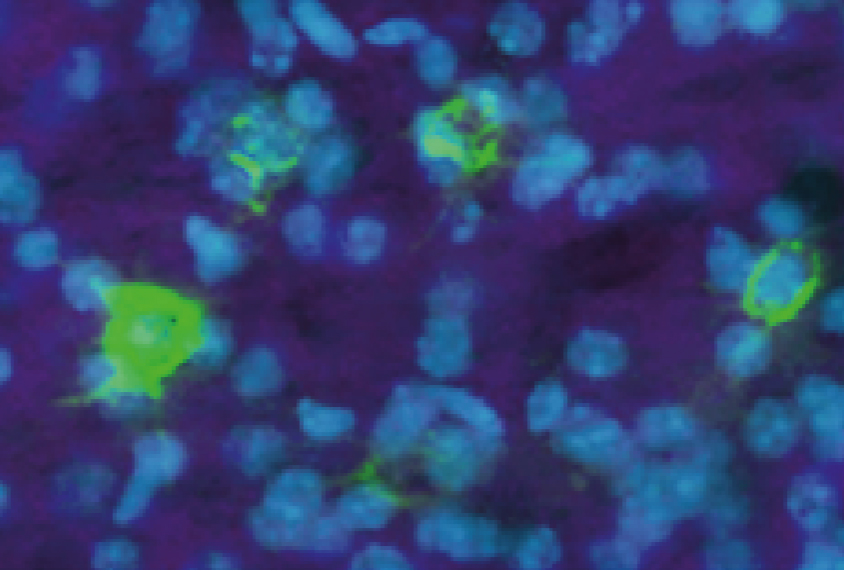
The researchers looked at mice lacking SHANK3, a gene found to be mutated in roughly 1 percent of people with autism. Using various techniques, the researchers found that these mice have less parvalbumin in their brains than do controls, as prior studies have shown.
But the researchers took another step, yielding results that upend conventional wisdom. They analyzed the mutant mice for a protein that coats the outside of parvalbumin interneurons. The marker showed up at similar levels in both sets of mice, suggesting that these cells are present in equal numbers in both.
Together, the findings suggest that the drop in parvalbumin levels in the mutant mice is the result of the interneurons in those mice producing abnormally low levels of parvalbumin, rather than from loss of the neurons.
Other researchers are cautious in interpreting the results or their implications for autism, however.
Several studies support the idea that loss of parvalbumin leads to lack of inhibition in brain circuits, notes Takao Hensch, professor of molecular and cellular biology at Harvard University. In that case, he says, either scenario — loss of the neurons or only of parvalbumin — would lead to a signaling imbalance. (Hensch has published some of the evidence suggesting that mouse models of autism have too few parvalbumin interneurons.)
Of mice and men:
It’s also unclear whether the findings from mice would hold up in people — and whether studies in people need to take this effect into account.
In a study published 27 February in Cerebral Cortex, for example, researchers stained postmortem brain tissue from 11 people who had autism and 10 controls for three types of inhibitory neurons2. The samples came from the prefrontal cortex — a region responsible for planning, reasoning and other higher-order functions.
The researchers saw fewer neurons stained with parvalbumin in the prefrontal cortex of individuals who had autism than in controls, but no difference in the two other neuron types. This finding supports the theory that alterations to parvalbumin neurons are important in autism.
However, it is not clear whether these findings result from a loss of parvalbumin or of the interneurons themselves, says Verónica Martínez-Cerdeño, assistant professor of pathology at the University of California, Davis, who led this study.
What’s more, low levels of parvalbumin — whether from a direct lack of the protein or of the cells that produce it — may have the same effect: A parvalbumin interneuron that lacks the protein might not be functional, says Manuel Casanova, ch, who was not involved in the work.
“The bottom line,” Casanova says, “is they are not doing their job.”
By joining the discussion, you agree to our privacy policy.