Glowing sensors shine new light on protein interactions
Researchers may soon be able to easily visualize protein pairings in living cells through vibrant flashes of color.
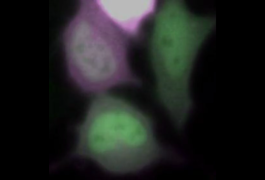
Researchers may soon be able to easily visualize protein pairings in living cells through vibrant flashes of color.

A new online resource called MouseID Genes stores information about gene interactions in the developing mouse brain and spinal cord.
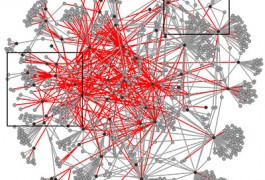
Researchers have mapped interactions between hundreds of variants of proteins made from known autism risk genes, they reported 11 April in Nature Communications. The highly connected network reveals that autism genes tend to interact with each other and with many more partners than suspected.

Researchers have created a network of various forms of many proteins linked to autism, revealing new molecular interactions that may play a role in the disorder. The unpublished work was presented in a poster last week at the Salk Institute, Fondation IPSEN and Nature Symposium on Biological Complexity in La Jolla, California.

The ten notable papers picked by SFARI staff describe superb contributions that span the breadth of autism research from molecules to behavior. But we recognize that ten other articles might have been selected without loss of enthusiasm or excitement.
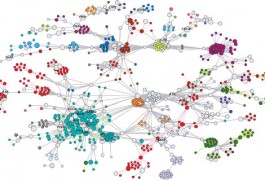
Researchers have developed a computational technique that allows them to simultaneously use multiple types of information — including gene expression, ratings and associations — to identify candidate genes for a disorder. The unpublished results were presented Sunday at the 2011 Society for Neuroscience annual meeting in Washington, D.C.

Researchers have identified hundreds of previously unknown connections between proteins involved in autism spectrum disorders, according to a report published last week in Science Translational Medicine.
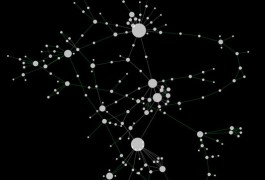
The pattern of interactions among different genes in yeast cells changes in response to disease-like conditions, in this case a DNA-damaging agent, according to a study published 3 December in Science. Mapping epistasis — how various cellular factors work together — is key to understanding complex disorders, such as autism.

Two independent teams have identified the genetic culprits of three rare, inherited diseases by sequencing the genomes of several members of the same family. As the cost of whole-genome sequencing plummets, this family-based approach will reveal candidate genes not just for rare diseases but for common, complex disorders such as autism, experts say.
To examine protein interactions inside brain cells, scientists typically zero in on one gene at a time. A new method described in today’s Nature simultaneously measures expression of the whole genome.