THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
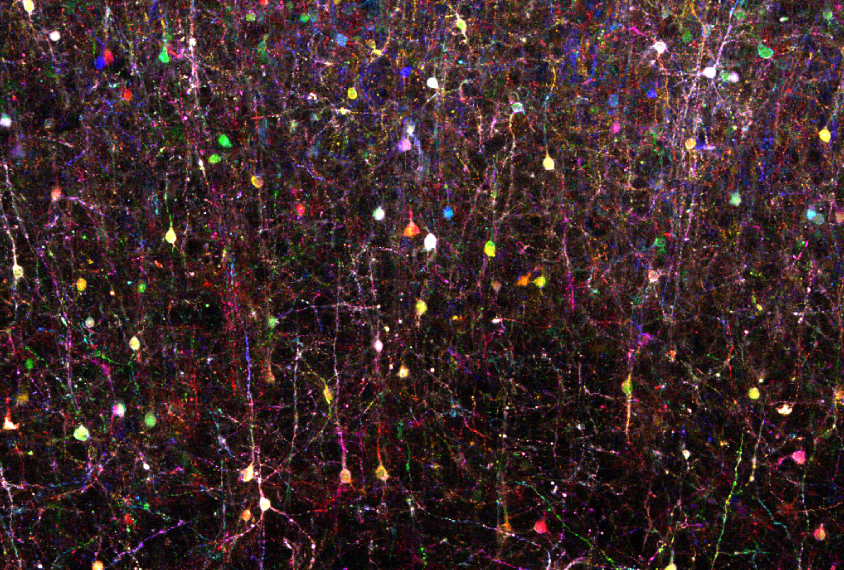
A team of scientists has applied ‘Brainbow’ — a popular tool for painting individual brain cells of lab animals in distinct colors — to the human brain. Researchers presented the stunning, Technicolor-like images today at the 2017 Society for Neuroscience annual meeting in Washington, D.C.
The findings could help researchers to map and characterize the cells that make up the human brain.
“There is actually no good catalog classifying human neurons,” says Shu-Hsien Sheu, a senior scientist in David Clapham’s lab at Janelia Research Campus in Ashburn, Virginia, who presented the findings. “We know that we probably have neurons that look different than neurons in mice, but they have never been well studied.”
The complex jumble of neurons and other cells in the brain are akin to a tub of tangled spaghetti, Sheu says. It can be difficult to trace individual pieces of ‘spaghetti,’ or cells. By painting each one a different color, researchers can follow the cells more easily.
Since 2007, researchers have been able to use Brainbow to dye individual cells of an animal’s brain. In this approach, researchers typically use viruses or genetic engineering to coax an animal’s brain cells to produce different combinations of up to four colored proteins. Depending on how much of each color a cell produces, researchers have access to a palette of more than 100 different hues.
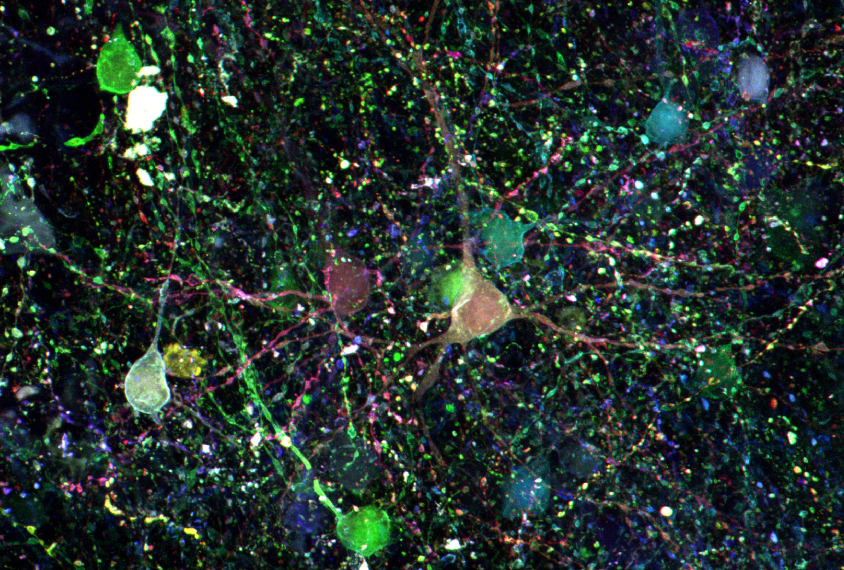
Dye job:
Sheu applied the virus-based version of the technique to samples of live brain tissue from people who had undergone surgery for epilepsy. He shaved the brain samples into ultra-thin sections and placed them in a culture dish with a nutrient-rich solution to keep the cells alive. He dribbled a few tiny droplets of a solution containing the viruses onto the surface of the slices, and incubated them for up to a week. He then examined the samples under a microscope, and saw a wash of different colors: The technique had successfully dyed multiple cell types.
“I was surprised to see it actually works,” Sheu says.
However, many cells in the samples had been damaged from seizures and degraded quickly over time. So Sheu could not detect the characteristic layered pattern of the cerebral cortex, the brain’s outer layer.
To obtain different types of brain samples, he collaborated with researchers at the Allen Brain Institute, who routinely collect live brain samples from hospitals in the Seattle, Washington, area. One member of the institute stands by during the surgeries so that samples of brain tissue can be immediately placed in a solution of artificial cerebrospinal fluid.
“The preservation of the tissue sample is a lot better,” Sheu says.
When Sheu and his colleagues applied the viruses to these samples, they were able to see the characteristic layered pattern of the cerebral cortex. The researchers are trying different types of viruses to see whether they can shorten the span of time it takes to dye the cells from seven days to under three.
“We want to minimize the culture span, because presumably these neurons change their shape over time, and we don’t want that to happen,” Sheu says. “We want to have a dataset that’s as close to the ones in the brain as possible.”
For more reports from the 2017 Society for Neuroscience annual meeting, please click here.
By joining the discussion, you agree to our privacy policy.