THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
A new technique enables scientists to use a pulse of light to control the activity of a single neuron1. The approach can reveal how cells in brain circuits coordinate.
Scientists can activate or silence neurons using optogenetics, an approach in which they genetically engineer neurons to carry light-sensitive proteins called opsins. These opsins make the cells respond to certain colors of light.
Light focused on small areas of brain tissue can limit the effects of the technique to a select group of cells. But it is tricky to target individual cells because neurons typically nestle within a web of fine projections — axons and dendrites — from nearby cells. Even a tightly focused light beam can illuminate and activate neighbors through those projections.
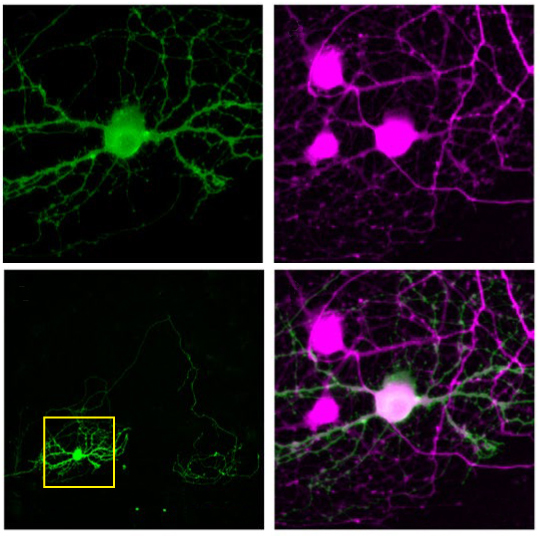
To avoid off-target activation, the researchers confined the light-sensitive proteins to each neuron’s cell body, the part that contains the nucleus. They achieved this by combining the genes for an opsin with those for a small fragment of another protein, the kainate receptor KA2 subunit, which appears only in the cell body. By engineering neurons to carry the linked genes, they created cells that produce a fused version of the two proteins.
The team tested the approach in cultured neurons. They recorded the neurons’ activity with an electrode and confirmed that only the cell targeted with a small spot of light fires. They were able to achieve the same result in slices of a mouse brain. They described their approach in December in Nature Neuroscience.
The researchers then demonstrated that the fused-protein approach can reveal how neuronal circuits function. They recorded the firing of a single cell in response to stimulation of each of the surrounding cells in slices of mouse visual cortex.
Some cells evoke a stronger response in the center cell than others do, an observation that could help scientists understand how circuits coordinate and modulate their activity.
By joining the discussion, you agree to our privacy policy.