THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.


Protein production is known to go awry in some forms of autism, and researchers are beginning to home in on the molecular players involved. For example, some people with autism have a genetic glitch that causes them to produce too much of a protein called EIF4E that can ramp up protein synthesis1.
Mice engineered to overproduce EIF4E end up with too much protein at synapses, the junctions between neurons. They also show altered communication between neurons, as well as repetitive behaviors reminiscent of those seen in autism2.
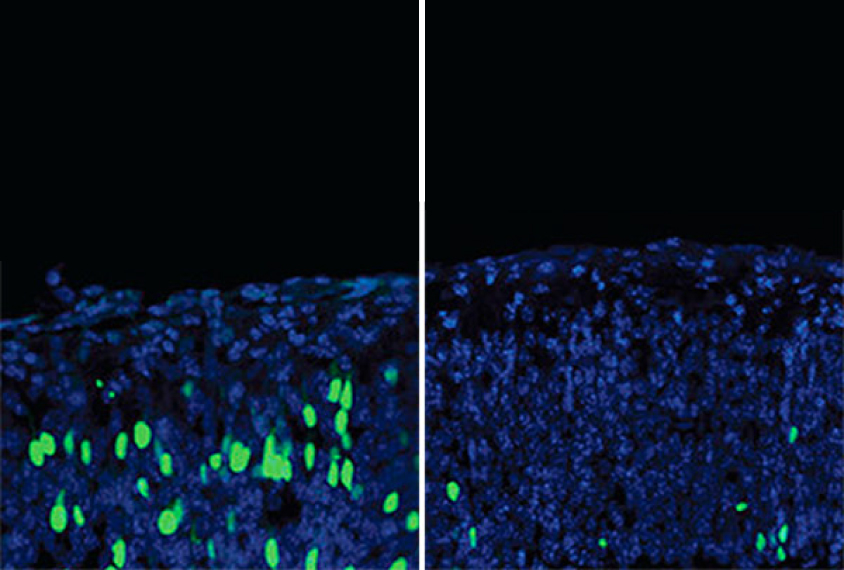
Freda Miller and David Kaplan, senior scientists at the Hospital for Sick Children in Toronto, Canada, have found a new role for EIF4E. They found that the gene acts early in brain development to ensure that neural stem cells mature into the right number of neurons3. Their evidence suggests that too little EIF4E unleashes an overproduction of new neurons in brain tissue from embryonic mice.
It’s not clear yet whether EIF4E’s role in the birth of neurons has anything to do with autism in people. But, intriguingly, some children with autism have large heads, a symptom that may reflect an overproduction of neurons in the brain. When embryonic mice are given a drug that causes the brain to make too many neurons, the animals later show social deficits and repetitive behaviors reminiscent of autism.
Miller presented data for EIF4E’s effects on the formation of neurons at the 2015 Wiring the Brain meeting at Cold Spring Harbor Laboratory in New York. We sat down with her and Kaplan to find out more about this early role for the autism candidate and whether there is any possibility of intervening.
SFARI.org: How did you become interested in autism genes that might influence neural development?
Freda Miller: When people started finding genes associated with autism, we looked at 60 of them to see if they were expressed in stem cells that give rise to neurons in the cortex, the brain’s outer rind. To our surprise, almost all of them were.
S: Why was this a surprise?
David Kaplan: Almost all of those autism-linked genes were thought to function after birth. But we found that many of them are expressed in embryonic neural stem cells, suggesting that they may have functions before birth.
S: What led you to focus on EIF4E in particular?
FM: It had been discovered that EIF4E is a part of various protein complexes that either initiate the translation of RNA into proteins or repress it. One of the repressive complexes includes FMRP, the protein that is missing in fragile X syndrome. Increasing EIF4E levels in adult mice also leads to autism-like behaviors. We decided to find out whether EIF4E is important to embryonic neural stem cell function and brain development.
S: How did you determine its role in brain development?
FM: We looked at all the repressive complexes that EIF4E belongs to and asked, ‘When we mess them up, does something happen?’ The one that jumped out at us was a complex involving EIF4E and another protein, called 4E-T. When we decreased the expression of either EIF4E or 4E-T in embryonic neural stem cells, we saw that there were too many neurons being produced at the wrong times during development. When we increased the expression of EIF4E, we saw too many stem cells and not enough neurons being formed.
This was really neat, given theories that some people with autism start out with too many neural stem cells that become neurons at the wrong times during development.
S: How do you think the EIF4E/4E-T protein complex regulates the development of neurons?
FM: The complex is basically an off switch for making proteins that tell stem cells to give rise to neurons. It works by binding to certain messenger RNAs (mRNAs) and preventing them from being translated into proteins. When EIF4E/4E-T itself is turned off, it stops binding to mRNAs. Those mRNAs then get translated into proteins that help make neurons. Too much EIF4E/4E-T activity results in two few neurons being made, and too little EIF4E/4E-T activity results in too many neurons being made.
S: Is there a way to prevent these glitches in neuron production?
DK: For the brain to develop normally, embryonic neural stem cells must become neurons at precisely the right time and place. Treatment is tricky. If the problem starts before birth, can you treat it? Maybe, since we have discovered drugs that can speed up or slow down neurogenesis. But when do you apply them, and how can we be sure that we are correcting the problem?
FM: I am an optimist about all of this. I think that one of the amazing things about the brain is that it is incredibly plastic at these developmental stages — meaning that even if you have some mistakes in setting up the number of neurons, you might be able to compensate for and correct those mistakes after birth. I believe that’s partly what behavioral therapy is doing — it’s compensating for earlier mistakes by enabling the correct rewiring of the brain. I think that these kinds of behavioral therapies, and potentially pharmaceutical treatments, could help us to build the appropriate brain circuits.
For more reports from the 2015 Wiring the Brain meeting, please click here.
References:
1. Neves-Pereira M. et al. J. Med. Genet. 46, 759-765 (2009) PubMed
2. Santini E. et al. Nature 493, 411-415 (2013) PubMed
3. Yang G. et al. Neuron 84, 723-739 (2014) PubMed
By joining the discussion, you agree to our privacy policy.