THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
Children with autism tend to carry variants in stretches of DNA that flank autism genes. These regions, once dismissed as ‘junk DNA,’ are now known to sometimes control the expression of genes that play key roles in brain development.
The study is small, with just 53 families that each include one person with autism. Still, the findings, published 7 January in the American Journal of Human Genetics, indicate that sequencing whole genomes could reveal the genetic cause of autism in as much as 30 percent of people for whom faster and cheaper sequencing methods come up short1.
“That there [may be] another significant genetic signal in the noncoding regulatory elements is really exciting,” says lead researcher Evan Eichler, professor of genome sciences at the University of Washington in Seattle. “It suggests another class of mutation, which gives us a lot of hope that sequencing whole genomes is going to be worthwhile.”
Eichler and his colleagues scoured sequences of entire genomes — genes and the regions between them — for previously unidentified variants that might be involved in autism. Prior attempts to sequence all the protein-coding regions of the genome, or exomes, had not turned up any autism-linked mutations in these 53 families. The family members also do not carry any large DNA deletions or duplications, called copy number variants (CNVs), known to be involved in autism.
Comparing the sequences of the individuals with autism and those of their unaffected siblings, the researchers found that people with autism are more likely to have genetic variants — either single base-pair changes in the sequence or small CNVs — in swaths of DNA abutting known autism genes. But the researchers only found the variants after they restricted their search to regions of the genome already implicated in autism, and even then the statistical significance is modest.
“[The researchers] see these intriguing signals near genes that we already know are involved in autism and are some of the most robust autism genes,” says Michael Talkowski, assistant professor of neurology at Harvard Medical School, who was not involved in the study. “It’s a start, and it’s a glimpse of what we might be able to find.”
Here be dragons:
Most genetic studies focus on exomes to find mutations, but regions between genes or within genes that are not made into protein can still be deeply involved in modulating gene expression. The difficulty is in knowing where these regulatory regions are.
Some studies have suggested that loosely packed regions of chromosomes are more likely than tightly wound ones to contain crucial regions. And parts of the genome that have been preserved across evolution — that is, unchanged across different species — are thought to serve important purposes.
Eichler and his team used these two criteria to look for autism-linked variants in 208 participants from the families. For 40 of the families, they also had genomic data from an unaffected sibling.
The researchers found that people with autism are no more likely than their unaffected siblings to have non-inherited, or de novo, variants in these probable regulatory regions. To increase their chances of finding a telltale variant, they then focused on 1,000 kilobase stretches on either side of 57 autism risk genes.
They found significantly more de novo variants among children with autism than among their siblings between 10 and 100 kilobases on either side of these genes. For example, they found six genetic variants in children with autism within 50 kilobases of the autism genes, but none in their unaffected siblings.
They also looked for CNVs and variants within autism-risk genes that previous sequencing methods might have missed. Whole-genome sequencing can pinpoint small CNVs, whereas traditional methods are usually limited to those larger than 100 kilobases.
Overall, 16 of the 53 people with autism have one or more of these rare variants, meaning they are present in a single person with autism or a single family; nine of the families carry CNVs or mutations that clearly alter genes.
“If in all of those families [these variants] turned out to be the cause of their autism, that would be a significant chunk of additional heritability of autism explained,” says Eichler.
Perfect storm:
In five of the nine families, the child with autism has multiple risk variants. For example, one girl inherited deletions in regions within and around ARID1B and SCN2A, two strong autism candidate genes, from her mother. She also inherited, from her father, a deletion in the coding region of a gene called CACNA2D4 that helps regulate neuronal activity.
Both parents are unaffected, but this “perfect storm” of mild risk factors may have caused the girl’s autism, Eichler says. Mutations in the regulatory regions may be milder than those found by exome sequencing, which often completely disrupt a gene, he says.
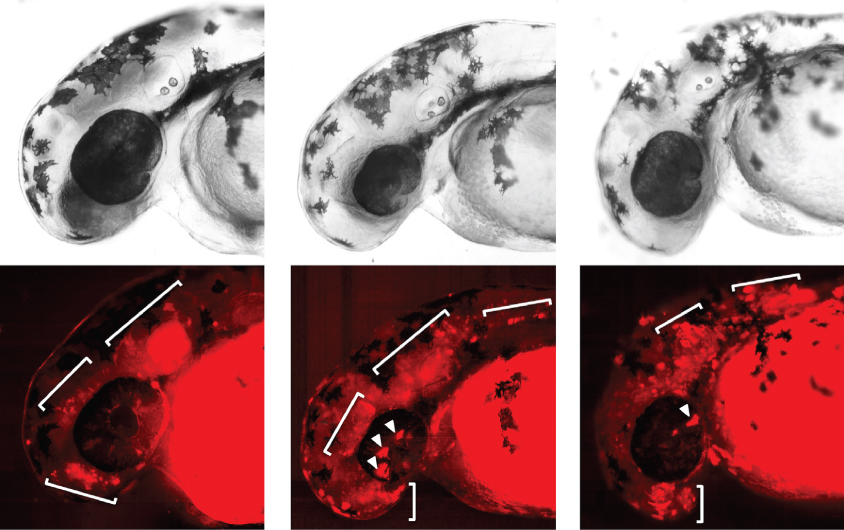
To confirm that a mutation affects a regulatory region, researchers need to monitor its effect on the gene’s function. In the study, they tracked the effect of one new de novo deletion in the noncoding region of an autism-linked gene called DSCAM. They fused portions of a region found to be deleted in DSCAM in one boy with autism to a gene that codes for a fluorescent protein and introduced this product into zebrafish. The presence of fluorescent proteins in the fish’s brains revealed that the region does indeed activate genes in neurons.
The findings, although preliminary, hint at the fruitful places to start looking in these new sequences, says Talkowski. “[The new study] is a tantalizing look at what a well-powered study might tell us,” he says. “It tells us the logical next step, and it’s a step we’re all very excited about.”
Look ahead:
Several efforts are underway to sequence more whole genomes from people with autism and their family members, including 2,000 new sequences set to be released later this month.
These sequences are likely to yield many new variants linked to autism, but finding a signal in the massive amount of data will not be easy, cautions Lucia Peixoto, assistant professor of medicine at Washington State University, who was not involved in the new work.
“This is a very promising study, but it shows the double-edged sword of whole-genome sequencing,” she says “There’s a lot of exciting things to come ahead, but it’s going to be harder to dig through it.”
The study also found variants in the exome that researchers didn’t find when they scanned exome sequences for deletions or duplications. It yielded sequences for nearly 2,000 genes usually missed by exome sequencing and identified nearly 7,000 new small CNVs that may be involved in autism.
“It’s increasing power even in areas that are supposed to be covered by whole-exome sequencing,” says Peixoto. “It seems that it’s clear that whole-genome sequencing will become the standard.”
By joining the discussion, you agree to our privacy policy.