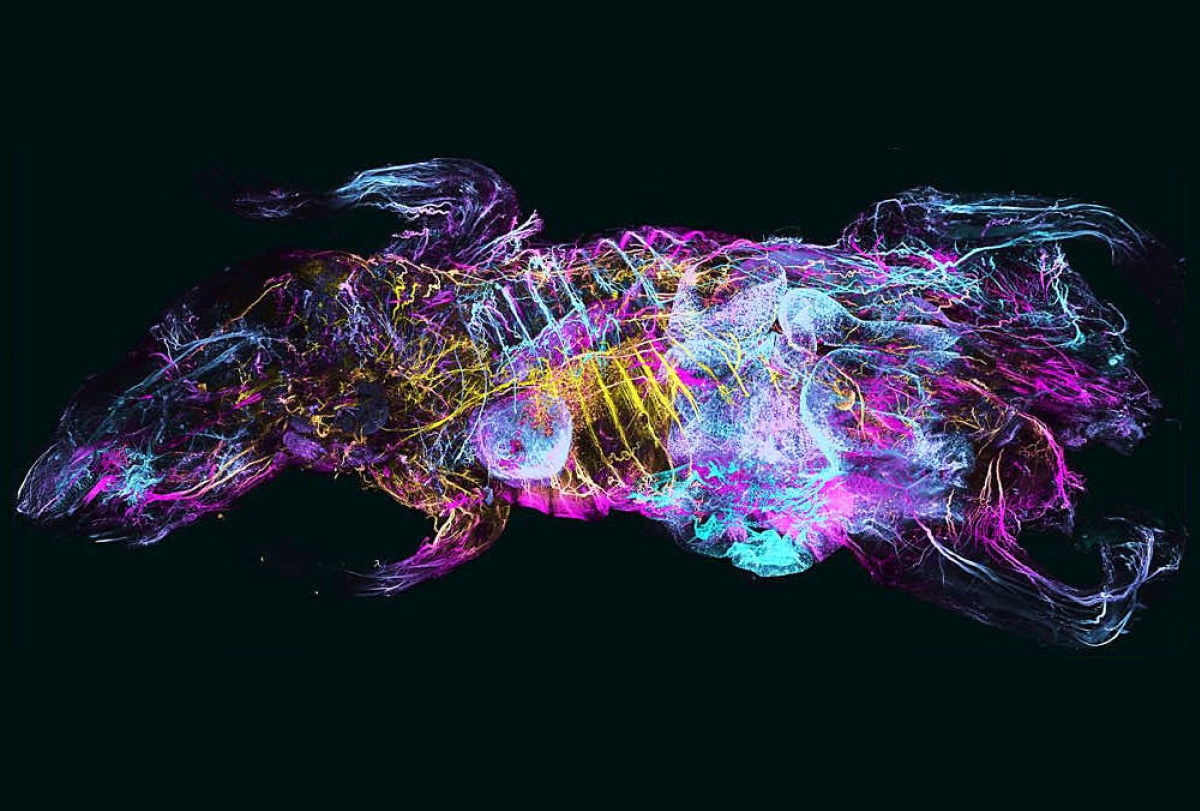
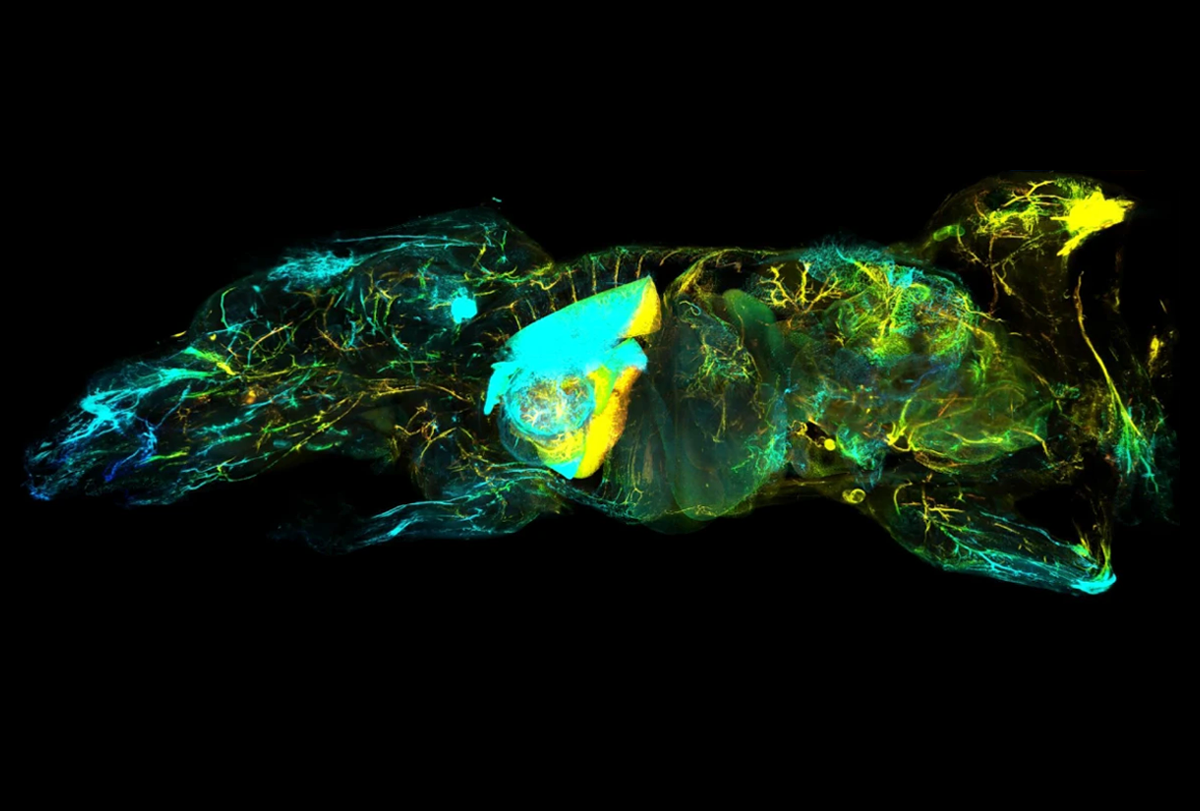
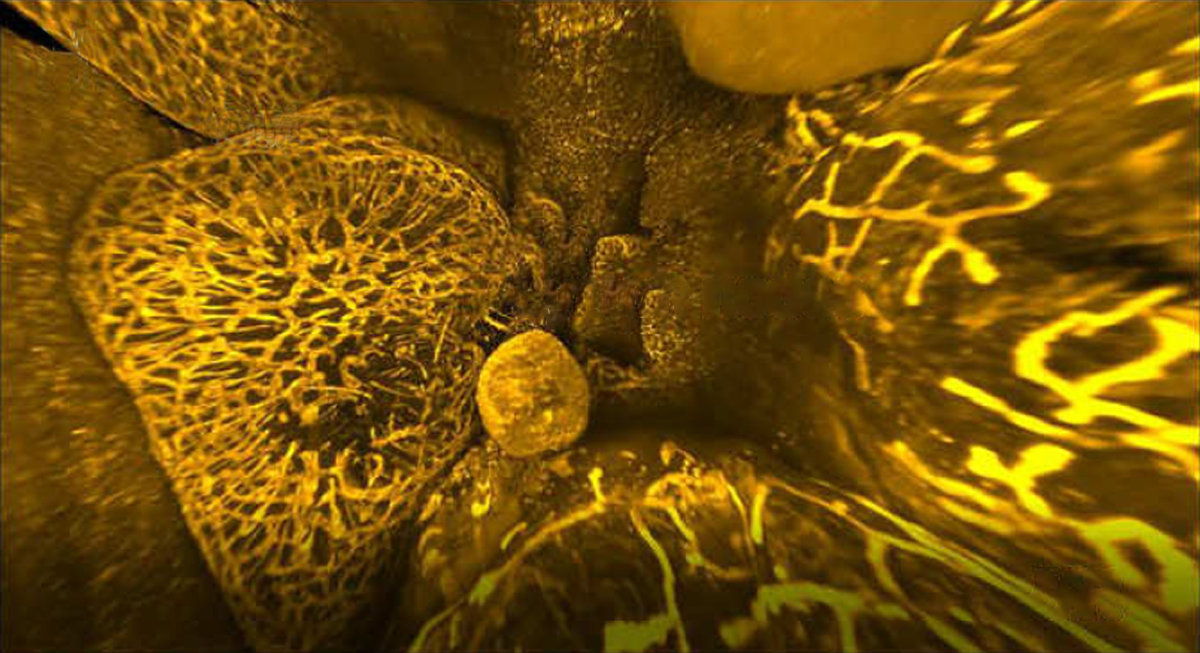
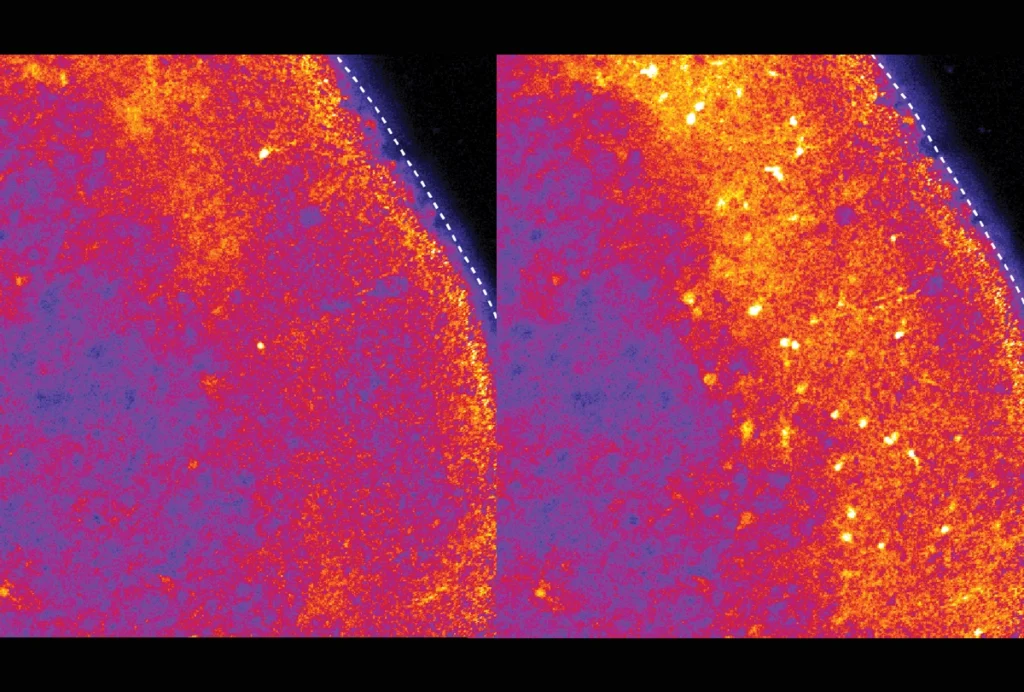
By turning mice transparent and using fluorescent antibodies to label specific cell types, researchers have created exquisitely detailed whole-body maps of intact neurons, immune cells, blood vessels and lymph networks.
The technology can help scientists understand how the nervous system interacts with other parts of the body, says lead investigator Ali Ertürk, director of Helmholtz Munich’s Institute for Tissue Engineering and Regenerative Medicine in Germany. “In the context of autism, it could provide a whole-body perspective of the neurological alterations associated with the condition.”
The new technique, known as wildDISCO and described last week in Nature Biotechnology, is the latest advance in tissue clearing, in which scientists bathe organs and even entire animals in chemicals postmortem to render them transparent — opening a view into specific cells or proteins tagged with fluorescent molecules.
wildDISCO enables researchers to tag many different types of cells across the roughly 2-centimeter-thick bodies of lab mice. It overcomes a key challenge inherent to previous tissue-clearing techniques such as CUBIC that depend on genetically modified rodents, which are expensive and time-consuming to create and only produce fluorescent proteins in a limited set of cells of interest, such as neurons.