Studies find high rate of rare new mutations in autism
Three new studies analyzing genetic data from families in which just one child has autism have found the strongest evidence yet that rare new mutations contribute to the disorder.
Charting the structure and function of the brain’s many circuits may unravel autism’s mysteries.

Three new studies analyzing genetic data from families in which just one child has autism have found the strongest evidence yet that rare new mutations contribute to the disorder.

Deletions on a segment of chromosome 11 are associated with autism, attention problems and obesity, according to a study published in the June issue of the American Journal of Medical Genetics Part A.

What’s known about the genetics of autism supports the ‘snowflake’ hypothesis — that the molecular underpinnings of disease are essentially unique from individual to individual — says human geneticist Brett Abrahams.

Mice with a mutation in SHANK3, a leading autism candidate gene, show moderate social defects, including less-than-normal interest in other mice. The findings, published 27 May in Cell, suggest that mutations in different sites on the gene can lead to different behaviors. This paper was retracted on 17 January 2013. Associate director of research Alan Packer discusses the implications of the retraction here.
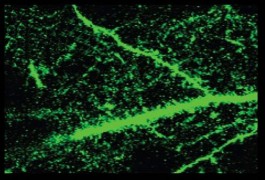
A new study provides the first functional link between the schizophrenia risk gene DISC1 and two candidate genes for autism. DISC1 significantly alters expression of NRXN1 and NRXN2 at key phases of development, according to a brief report in the June issue of Molecular Psychiatry.
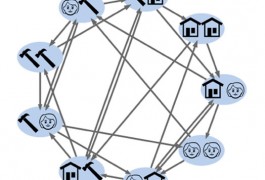
A complex mathematical technique can improve the sensitivity of experiments that rely on brain imaging, allowing researchers to study how the brain responds to sequences of stimuli, according to a study published in the June issue of NeuroImage.
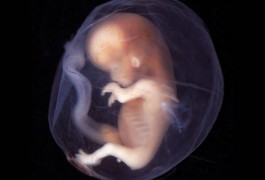
The placenta regulates the levels of the neurotransmitter serotonin in the brains of mice at a key stage in embryonic development, according to a study published 21 April in Nature. The results suggest that the fetal environment can influence the long-term mental health of children, including whether they later develop autism or schizophrenia.

A compound that activates a pathway related to learning and memory can enhance pair-bonding between prairie voles, according to a study published 7 April in Biological Psychiatry. Enhancing social learning — an individual’s response to social cues — during development could help treat autism.
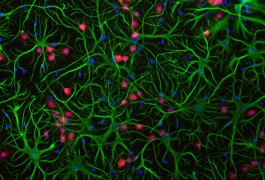
Harmful mutations in a gene that regulates the chemical environment outside of neurons are associated with both autism and epilepsy, according to a study published 31 March in Neurobiology of Disease.