Promising treatment
Families affected by fragile X syndrome can let out a modest cheer this week: the largest-ever randomized trial of a drug to treat the syndrome has just cleared its second phase.

Families affected by fragile X syndrome can let out a modest cheer this week: the largest-ever randomized trial of a drug to treat the syndrome has just cleared its second phase.
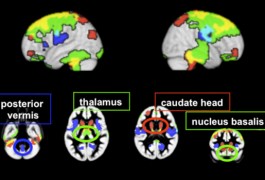
Children with fragile X syndrome show abnormal growth in several brain structures during the first few years of life, according to the first study to track how the disease unfolds in the brain during early development.

A decade of research on the biology of autism, combined with a steady rise in diagnoses, has finally piqued the pharmaceutical industry’s interest in developing drugs for the disorder. Preliminary data from one small clinical trial already show positive results, and results from several others are expected early this summer.

Moving a drug from the lab bench to the pharmacy’s shelves takes about 10 years. But for one controversial autism treatment, the process might be much quicker.

The latest findings on oxytocin — a.k.a. the ‘trust hormone’ — secure its position as a frontrunner among emerging treatments for autism.
A class of medications widely used during pregnancy to treat asthma and prevent early labor increases the baby’s risk of autism and other psychiatric disorders, according to a controversial review in the American Journal of Obstetrics and Gynecology.

There’s good news for autism research tucked into President Obama’s 2011 budget proposal: $222 million of it, to be exact.

Genetic variations that tweak the brain’s release of oxytocin — a hormone involved in social bonding and establishing trust — may increase the risk of developing autism or traits of the disorder, according to three new studies published in the past few months.

A large clinical trial to test the first drug specifically designed to treat autism is under way at 12 sites across the United States.

In the spirit of Thanksgiving, those dealing with autism received some welcome news last week: The U.S. Food and Drug Administration approved Abilify (aripiprazole) as a treatment for autism-related irritability.