Angelman syndrome
UBE3A’s link to synaptic pruning bolstered by fly study
Increasing or reducing the levels of the UBE3A gene, which is associated with autism and autism-related syndromes, results in altered patterns of synaptic pruning — a process that snips away brain cell connections.
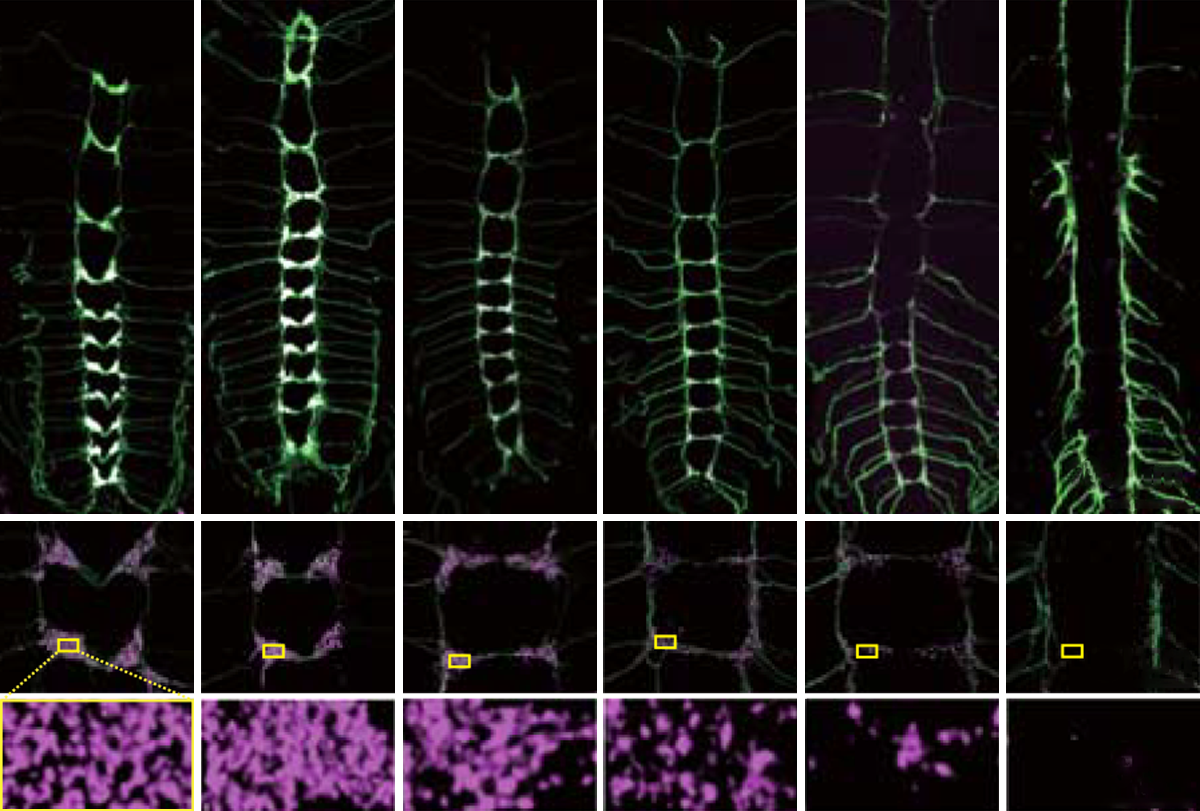
UBE3A’s link to synaptic pruning bolstered by fly study
Trials test utility of EEG biomarkers for autism-related conditions
This month’s Going on Trial newsletter dives into an electroencephalography biomarker that could track the efficacy of treatments for dup15q and Angelman syndromes, among other drug development news.
Trials test utility of EEG biomarkers for autism-related conditions
Is excess brain fluid an early marker of autism?
Brain scans of hundreds of infants suggest that up to 80 percent of those with autism have unusual amounts of cerebrospinal fluid. Researchers are studying how this might contribute to the condition.
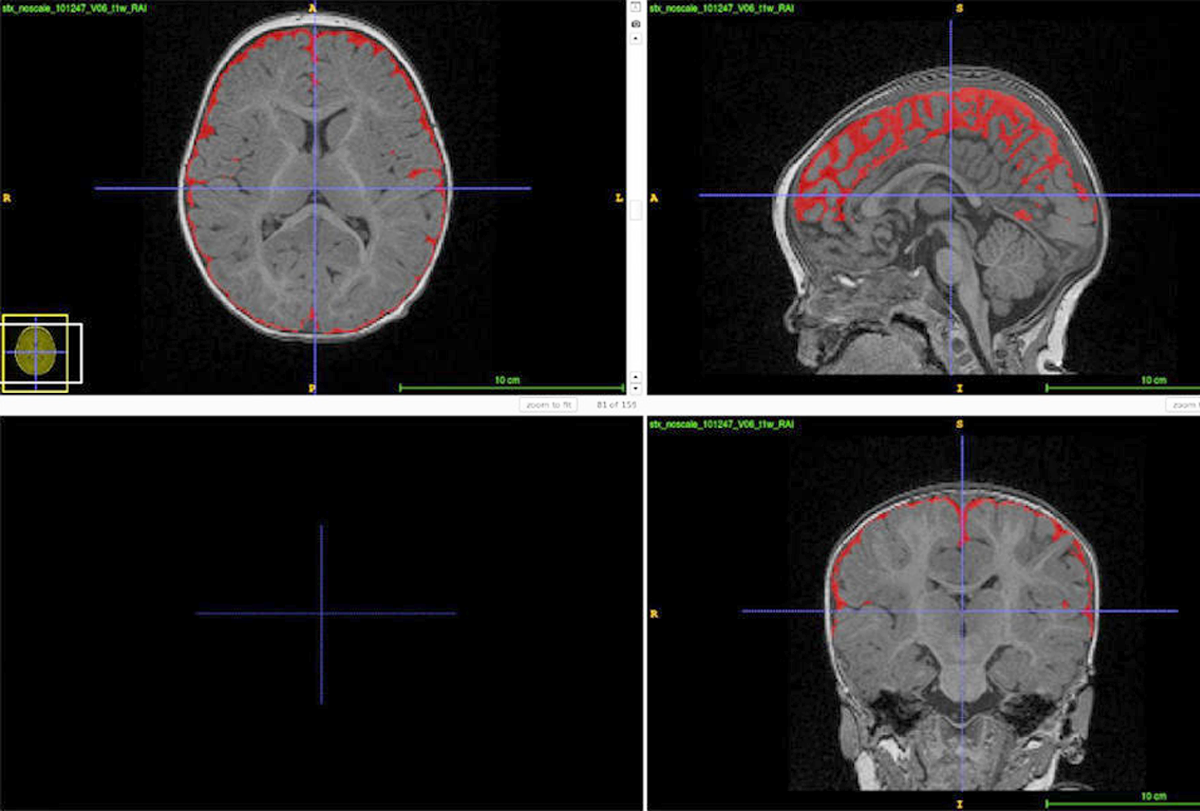
Is excess brain fluid an early marker of autism?
‘Gain-of-function’ mutation spawns autism traits
The mutation increases the activity of an autism-linked protein and leads to social difficulties and other behavioral differences in mice.
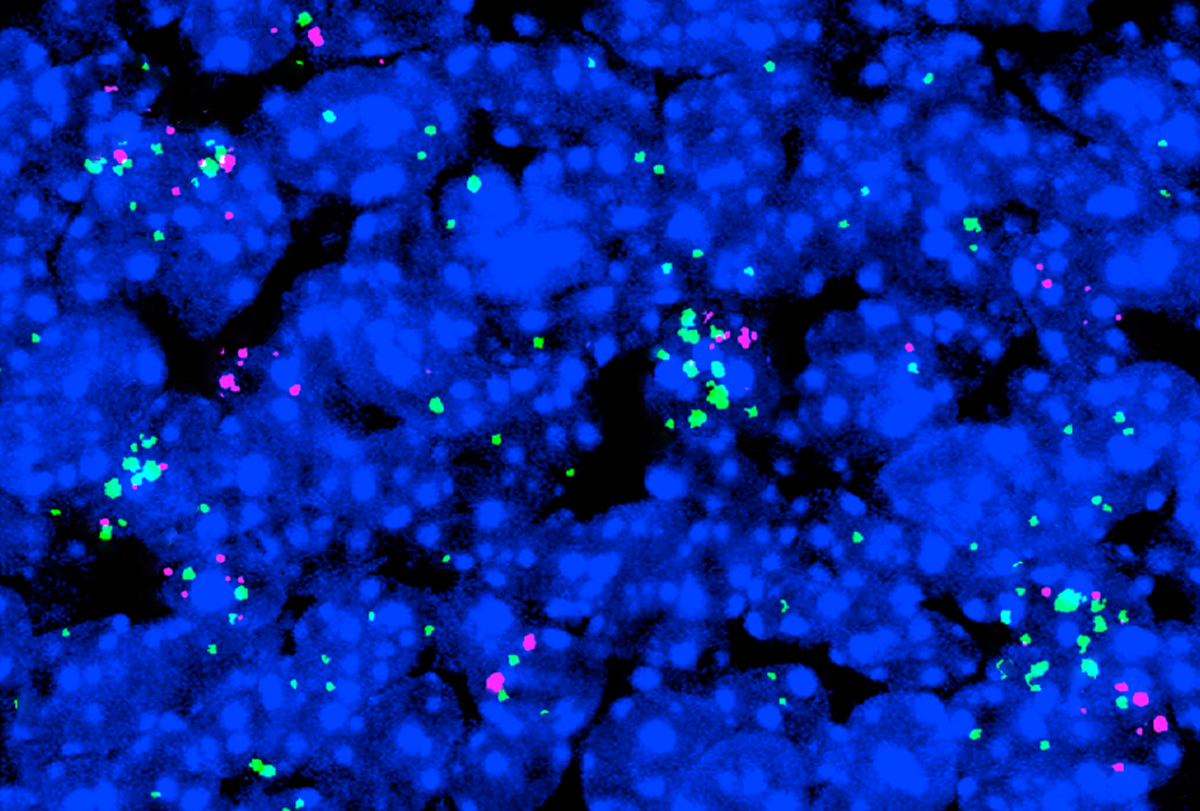
‘Gain-of-function’ mutation spawns autism traits
Prenatal exposures; Angelman trial suspension; autistic adult well-being
This month’s issue of the Null and Noteworthy newsletter breaks down some negative results involving prenatal exposures, an experimental treatment for Angelman syndrome, and the role that age at autism diagnosis plays in subsequent outcomes, and more.

Prenatal exposures; Angelman trial suspension; autistic adult well-being
Spotted around the web: Synthetic embryos; Angelman gene therapy
Here is a roundup of news and research for the week of 26 June.
Spotted around the web: Synthetic embryos; Angelman gene therapy
The future of autism therapies: A conversation with Lilia Iakoucheva and Derek Hong
If a therapy for autism’s core traits makes it to market, it will likely take one of three forms, the researchers say.

The future of autism therapies: A conversation with Lilia Iakoucheva and Derek Hong
RNA therapy restores gene function in monkeys modeling Angelman syndrome
The result raises hopes for an ongoing clinical trial in people — and offers fresh insight into the biology of imprinting and the UBE3A antisense transcript.

RNA therapy restores gene function in monkeys modeling Angelman syndrome
Single gene insufficient to account for dup15q, Angelman traits
UBE3A, a key gene associated with both autism-linked conditions, can explain most — but not all — of the syndromes’ atypical neuronal properties.
Single gene insufficient to account for dup15q, Angelman traits
Adult Angelman mice get some benefit from boosting UBE3A gene expression
The treatment eases the animals’ sleep troubles, suggesting it has clinically meaningful effects beyond what was thought to be a critical window in early life.
Adult Angelman mice get some benefit from boosting UBE3A gene expression
Explore more from The Transmitter
Cocaine, morphine commandeer neurons normally activated by food, water in mice
Confirming a long-held hypothesis, repeated exposure to the drugs alters neurons in the nucleus accumbens, the brain’s reward center, and curbs an animal’s urge for sustenance.
Cocaine, morphine commandeer neurons normally activated by food, water in mice
Confirming a long-held hypothesis, repeated exposure to the drugs alters neurons in the nucleus accumbens, the brain’s reward center, and curbs an animal’s urge for sustenance.
X chromosome inactivation; motor difficulties in 16p11.2 duplication and deletion; oligodendroglia
Here is a roundup of autism-related news and research spotted around the web for the week of 6 May.
X chromosome inactivation; motor difficulties in 16p11.2 duplication and deletion; oligodendroglia
Here is a roundup of autism-related news and research spotted around the web for the week of 6 May.
Decoding flies’ motor control with acrobat-scientist Eugenia Chiappe
The tiny performers steal the show in Chiappe’s sensorimotor-integration lab in Lisbon, Portugal.

Decoding flies’ motor control with acrobat-scientist Eugenia Chiappe
The tiny performers steal the show in Chiappe’s sensorimotor-integration lab in Lisbon, Portugal.