THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.

It’s been more than a century since German neuroanatomist Korbinian Brodmann mapped 52 distinct regions onto the brain’s surface. Brodmann painstakingly detailed the types of cells in each region and their arrangements — a mammoth effort at the time.
Since then, researchers have used imaging techniques to construct a panoply of new maps of the brain’s structure and function. But because of the human brain’s sheer size, researchers have been unable to detail it as they have done for mouse and monkey brains. The relatively low resolution of human brain maps has made it difficult to connect specific cells and neuronal projections, or axons, to large-scale patterns of brain activity and connectivity.
A team of researchers at the Allen Institute for Brain Science in Seattle have filled this void1. By combining two magnetic resonance imaging (MRI) techniques with high-resolution microscopy, they have created the most in-depth portrait to date of a single human brain.
We asked the project’s leader, Ed Lein, how his team put together the new atlas, and how they envision others using it.
Spectrum: Why did you create this atlas?
Ed Lein: Methods and standards for making brain atlases for model organisms have advanced tremendously in recent years. Available human brain atlases lack many of the features of these animal maps, however, and some atlases still in use are almost 100 years old. We needed a modern atlas that allows us to move from large structures all the way to individual cells. The human brain is enormous, so just generating the data is exceptionally difficult. And then we still need to interpret the anatomy.
We wanted to provide comprehensive anatomical coverage at microscopic resolution using multiple stains to delineate all of the different structures of the brain. Finally, we wanted to make this an open-access, online tool that allows researchers to start with a low-resolution view of the structures and zoom all the way in to the level of individual cells.
S: How did you make your atlas?
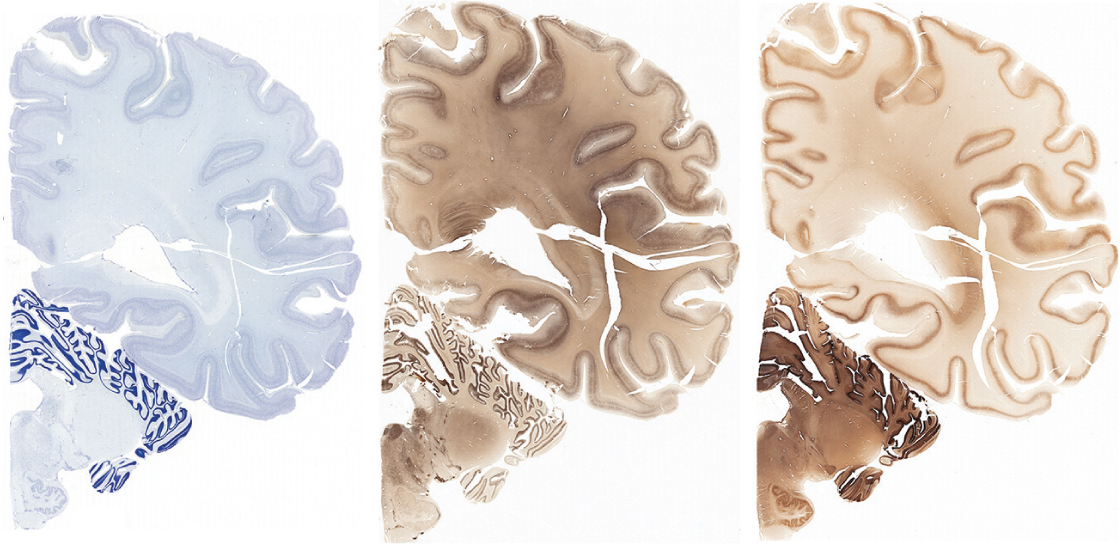
EL: First, we scanned the brain’s overall structure using magnetic resonance techniques. After that, we cut the brain into slabs from front to back, each approximately 1 centimeter thick. We then sliced each slab into 50-micron sections — about the thickness of a human hair.
We mounted these sections onto microscope slides and then we used a set of stains to label individual cells and their parts. A Nissl stain, for instance, allows you to see every single cell. We also used a stain that labels cells containing a protein called parvalbumin, a marker for a type of neuron that dampens brain activity, and a stain that labels another type of neuron and their axons and dendrites.
Because the sections are so big, we developed a new microscope platform that allows us to take many high-resolution images and digitally stitch them together. Then we identify and label brain regions using a couple of techniques. One is based on landmarks on the surface of the brain, and the other on the distribution and characteristics of cells in a particular region.
We drew the boundaries on the digital images so that anyone who uses the atlas can see what we’re calling a particular structure. And because they’re digital images, researchers could in principle relabel brain areas based on their own data.
S: How long did this project take?
EL: It took about four years. In the first year and a half, we optimized the techniques for slicing the brain, staining the slices, scanning them at high resolution and then digitizing them.
What took longer was the labeling process. We developed a nice strategy for doing this. Song-Lin Ding, a terrific anatomist, really drove the process. Once we got images of the brain slices, we printed them on posters. Ding identified each brain region on these posters and then handed his notations over to a team of graphic artists, who transposed the boundaries onto the digital images. We have stacks and stacks of these poster prints.
S: Can the information in this atlas be tied to other data the Allen Institute has collected?
EL: Yes, absolutely. One of our goals with this atlas was to make it useful for the wider brain imaging community. The resolution of functional neuroimaging is relatively low, which makes it difficult for researchers to identify which brain areas are showing functional responses. We performed MRI on the brain before we sliced it, and that allows us to correlate the cell types and boundaries we identified with the overall structure of the brain.
We have also linked the new atlas to our previously published genetic information. We have an atlas of the activity of all genes in finely dissected regions of the adult brain. For that, we had looked at about 1,000 regions across the human brain in a small number of individuals. These structures are in our new reference atlas, so you can go in and look at their genetic signatures and understand what makes each of them distinct.
We also have maps of the activity of single genes in various parts of the brain. This allows you to see exactly which structures and cells are using that gene.
The idea is to provide a map of the brain that we can layer other information onto, to begin to understand the nature of the structures that make up the functional brain.
S: How does your atlas compare with the one the Human Connectome Project released in July?
EL: Their effort is fantastic. They took a different approach, using several noninvasive imaging techniques to understand the common architecture and function of the human cerebral cortex across many individuals. This sort of atlas is probabilistic — it represents the average across many individuals rather than any single individual.
By contrast, structural atlases like ours focus on a small number of individuals, but in great, great detail. An obvious next step is to see to what extent the large-scale subdivisions in the Human Connectome map agree with the fine-scale cellular subdivisions in our map. The more that we can match these atlases up, the more useful they will be for the neuroimaging community.
By joining the discussion, you agree to our privacy policy.