THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.

Ever since Cagla Eroglu was a postdoctoral fellow, she’s been fascinated by star-shaped brain cells called astrocytes. In 2008, she set up a lab at Duke University in Durham, North Carolina, to investigate the role of astrocytes in forming neuronal junctions, or synapses.
Last year, Eroglu and her colleagues revealed that the relationship is reciprocal: Contact with synapses helps to give astrocytes their complex shape1.
The team also found that astrocytes express neuroligins, synaptic genes known to be mutated in some people with autism. Neuroligins help control astrocyte growth and shape in concert with proteins on neurons called neurexins that are also associated with autism.
Eroglu found that lowering neuroligin expression in astrocytes makes the brain less excitable. This result may be relevant to autism, which is thought to be characterized by too much excitation in the brain.
We talked with Eroglu, associate professor of cell biology and neurobiology, about how her findings advance the understanding of autism.
Spectrum: What motivated your latest experiments?
Cagla Eroglu: I have long investigated how astrocytes drive the formation of synapses in the mammalian brain. With this particular project, we wanted to turn the tables and ask: How might synapses drive the complex development of astrocytes?
S: In what way is astrocyte development complex?
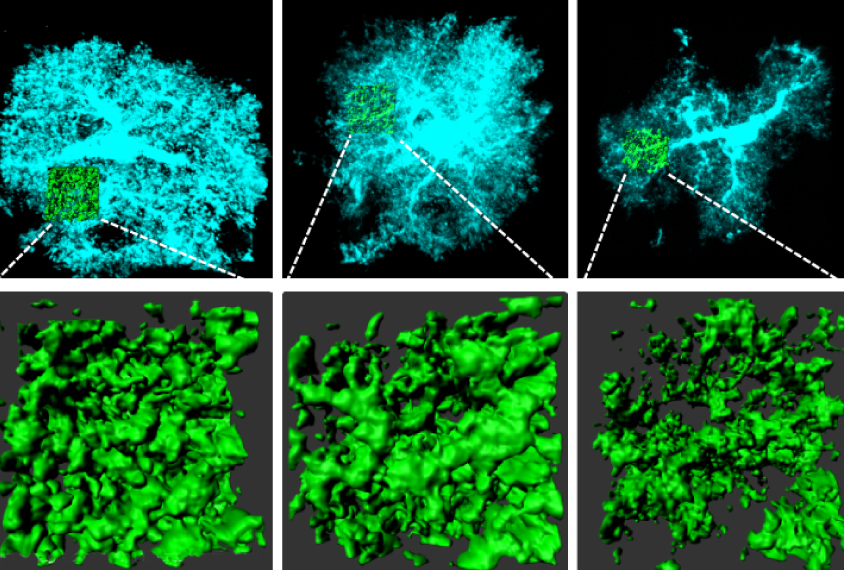
CE: Astrocytes have a unique structure that resembles a tree with many branches. You have main branches that give rise to littler branches and then leaves. In a mouse brain, one astrocyte’s branches can contact up to 100,000 synapses. A human astrocyte can contact up to 2 million synapses. It is a really incredible complexity.
It makes for a very beautiful cell, but it also gives that astrocyte the ability to ‘listen’ to the activity of many synapses at once. We wanted to understand how that amazing morphology forms. We hypothesized that interaction with neuronal elements could drive the morphology.
S: How did you test your hypothesis?
CE: We developed a cell-culture system in which we eliminated the other types of cells from the brain until we only had astrocytes and neurons. We found that intact but nonfunctioning neurons could trigger astrocytes to develop their complex shape.
The neurons didn’t need to be alive, so that gave us a clue that the complexity of astrocytes requires physical contact between these cells and neurons. We then realized there must be ‘sticky’ proteins on the surface of astrocytes that can bind to proteins on neurons, and potentially at their synapses.
S: What were these ‘sticky’ proteins?
CE: One protein family called neuroligin emerged as a strong candidate. Neuroligin has been studied almost exclusively in the context of neurons. But some work suggested neuroligins are also expressed in astrocytes. This made us curious: What are they doing in astrocytes? And are these the sticky proteins that connect neurons to astrocytes, and that drive astrocytes’ morphology?
We found three members of the neuroligin family that are expressed in astrocytes from rats and mice. We then used RNA molecules to suppress the expression of the genes — and got a very simple astrocyte, one with far fewer branches than usual. Suppressing one of these genes at a time produced an intermediate effect on the astrocyte’s shape, suggesting that all three proteins are essential for the cells’ complex shape.
However, it turns out the genes are not sufficient for astrocytes’ complexity.
We tamped down the expression of proteins called neurexins in neurons that we thought astrocyte neuroligins might interact with. The astrocytes in that culture were less complex. We also replaced neurons with tiny, thread-like materials called nanofibers that resemble neural networks, and covered the nanofiber scaffold with neurexins. These scaffolds could trigger the formation of complex astrocytes.
These experiments told us that there are two pieces to the astrocyte puzzle: neurexins in neurons and neuroligins in astrocytes. Together, these produce astrocytes in their typical form.
S: Is there any particular neuroligin that seems more important than the others?
CE: Yes, neurologin 2. We found that when we lowered the expression of NLGN2 in some astrocytes, the neuronal elements that touch those astrocytes have far fewer excitatory synapses. And when we decreased the expression of neuroligin 2 in about half of the astrocytes but not in neurons, we found a decrease in excitation and an increase in inhibition.
So tamping down neuroligin 2 in astrocytes shifts the balance in the brain toward less excitation and more inhibition.
S: Is this relevant to autism?
CE: Researchers have proposed that an imbalance between excitation and inhibition is a common feature of autism models and people with autism. Maintaining a balance between these two types of neuronal activity is crucial for proper brain function. Our finding is telling us that manipulating gene function in astrocytes is sufficient to change this balance.
Anyone who is studying the function of an autism gene should consider that a gene’s effects in neurons may not fully account for what they see in their autism models. If researchers are focusing only on neurons, and neglecting astrocytes, they may not be getting the full picture.
By joining the discussion, you agree to our privacy policy.