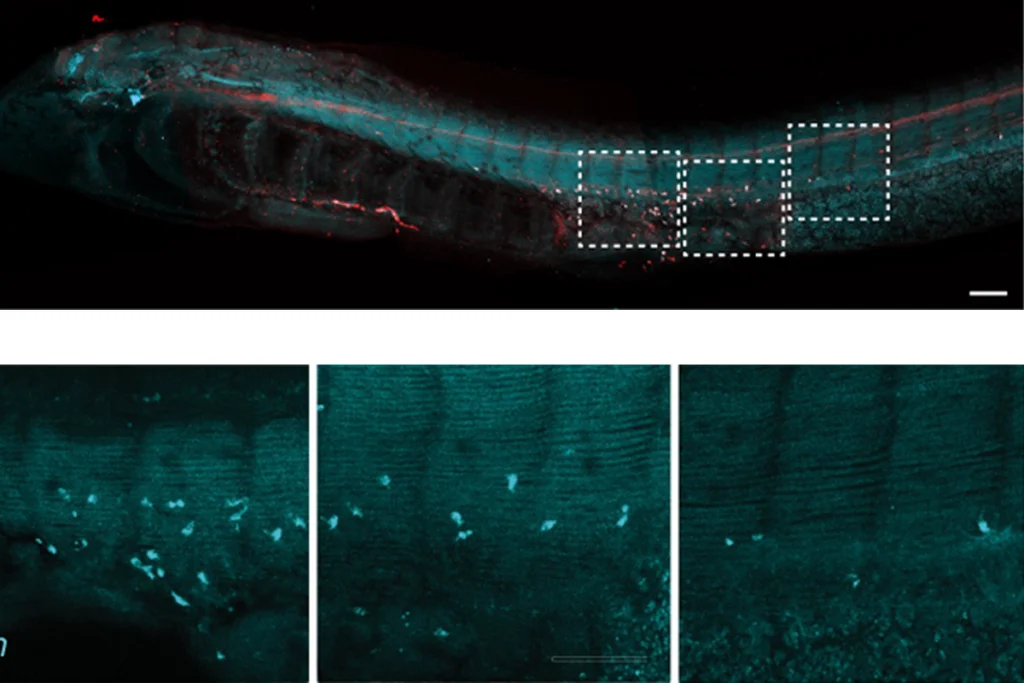
Wiring map reveals how larval fruit fly brain converts sensory signals to movement
The map diagrams more than half a million neuronal connections in the first complete connectome of Drosophila and holds clues about which brain architectures best support learning.
Feedback loops and layered pathways power the larval fruit fly brain, according to an analysis of the organism’s first complete wiring map, published today in Science.
The connectome traces all 3,016 neurons and 548,000 synapses in the central nervous system of a 6-hour-old female Drosophila melanogaster — from the axons that deliver sensory input to the brain, to the dendrites on neurons that send out brain signals to control movement.
“The connectome generates a lot of hypotheses,” says lead investigator Marta Zlatic, research leader at the MRC Laboratory of Molecular Biology at the University of Cambridge in the United Kingdom. “It gets us from a place where we were sort of in the dark, we were sort of guessing how neural computations are implemented, and now we have the structural candidates that could be implemented.”
The new wiring map is the fourth and most complex complete connectome reported to date: The first three — for the nematode Caenorhabditis elegans, the larval sea squirt Ciona intestinalis and the marine worm Platynereis dumerilii — detail just a few hundred neurons each.
Even at the larval stage, fruit flies have more complex behaviors than any of those organisms, says Shinya Yamamoto, assistant professor of molecular and human genetics and neuroscience at Baylor College of Medicine in Houston, Texas, who was not involved in the work. They move toward the smell of food and away from light, for example — actions that require brain structures that can convert sensory information into movement.
“It’s a ‘wow,’” he says of the new map. “Now we know by single-cell resolution how these things could actually work.”
Researchers have generated larger but incomplete connectomes for more complex brains, including a map of more than 20 million connections among 25,000 neurons in the adult fruit fly.
The new work is a “significant contribution” toward attempting to understand fundamental principles of information processing in the brain, says Gerald Rubin, executive director of the Howard Hughes Medical Institute’s Janelia Research Campus in Ashburn, Virginia. Rubin led the work on the adult fruit fly connectome but was not involved in the new study.
“This paper takes on that challenge and presents some nice ways of looking at it,” Rubin says. “None of them are the answer to how the brain works, but they’re different ways of thinking about how it might work.”
Z
Among the synapses, 66.6 percent are axons connected to dendrites, 25.8 percent are axons connected to themselves, 5.8 percent are dendrites connected to themselves and 1.8 percent are dendrites connected to axons.
Clustering neurons based on the kinds of connections they make revealed 93 neuron types. Neurons within each type share a similar physical structure, according to an analysis using an independent algorithmic tool, suggesting that neurons are best distinguished by connectivity rather than structure or function, Zlatic says.
Less than 15 percent of the neurons receive only one type of sensory input, the team found via an algorithm that uses the number of connections among cells to predict how signals flow through the brain. Dopaminergic neurons — which are involved in learning — received input from all senses.
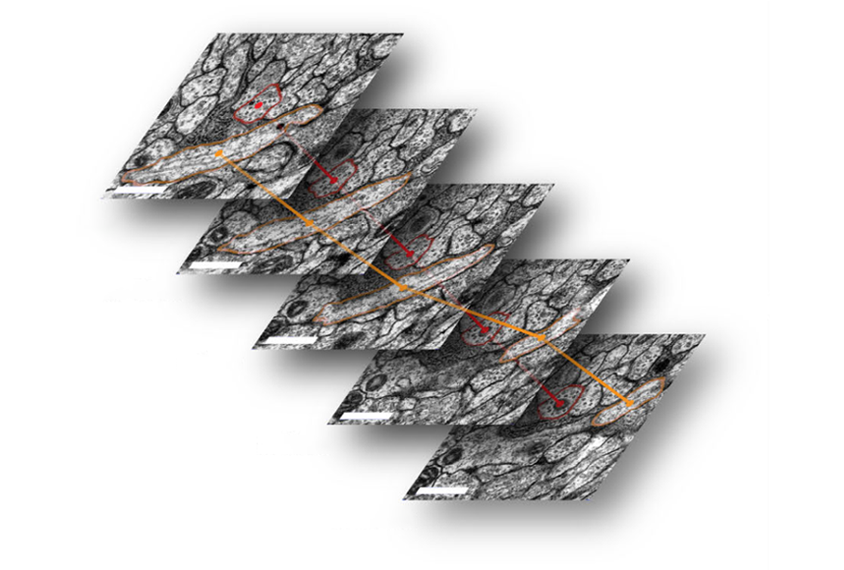
The path from the brain’s sensory input to its motor output is sometimes as short as two synapses, but layered on top of those short pathways are longer ones, spanning up to eight connections. The overlap creates a “nested” structure that likely gives the brain more computational power from fewer neurons, says study investigator Michael Winding, a postdoctoral research associate in Zlatic’s lab. “None of us expected this at all.”
Though most information flows from sensory input to motor output, signals also travel in the reverse direction, creating feedback loops between brain areas. And individual neurons send signals backward and forward through two-way, or recurrent, connections, the researchers found.
Although 41 percent of all neurons have recurrent connections, the number varies widely among neuron types: Almost no projection neurons or Kenyon cells — a type of neuron in the fly’s learning center — are recurrent, whereas 57 percent of dopaminergic neurons are.
“That’s really quite nice because we know that recurrent neural networks are pretty powerful in artificial intelligence,” Zlatic says. The findings reveal “what kinds of architectures are the best for learning.”
T
New electron microscopes can image an entire fruit fly brain in weeks. And software can trace neurons in a fraction of the time it takes researchers to do it manually, Winding says.
That will enable autism researchers to study genes linked to the condition in a more targeted way, Yamamoto says — particularly because many of the genes are essential for synapses to function.
“Having a synaptic-level map is what is necessary to understand what these genes do when mutated,” he says.
Zlatic says she plans to use optogenetics to create activity maps of the larva brain to understand how the connections function during different behaviors.
Explore more from The Transmitter

$278 million cut in BRAIN Initiative funding leaves neuroscientists in limbo

Reporting bias widespread in early-childhood autism intervention trials