THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
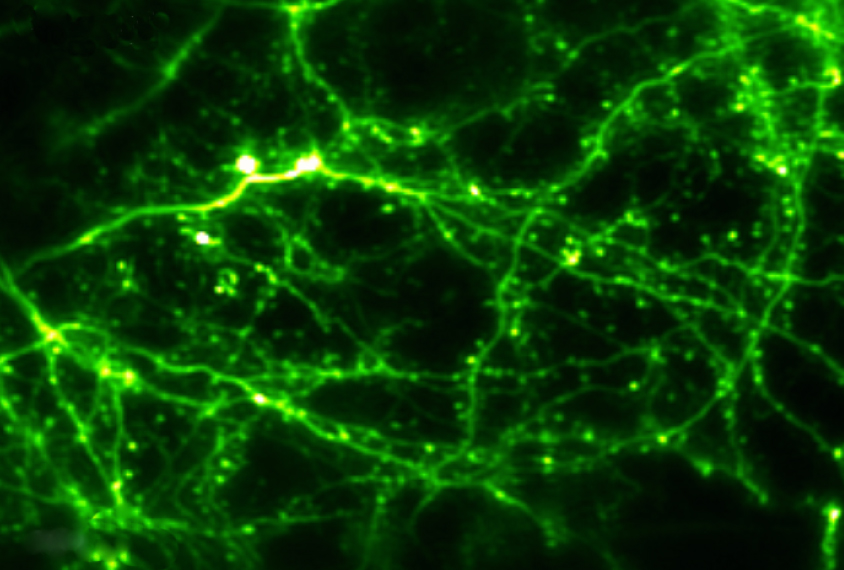
Live imaging techniques allow researchers to watch neurons in action and see how their firing patterns track with behaviors. In these methods, researchers typically engineer animals to produce proteins that fluoresce when neurons fire. They can then watch the bursts of fluorescent light through a specialized microscope aimed through a window in the skull.
Scientists have used live imaging to observe networks of neurons firing in mice as they move. Four modifications to the approach expand its use to monkeys, interior brain regions, fine cellular features and broad regions of the brain.
Clear view:
One new strategy, described 9 March in Neuron, provides clear pictures of neurons firing in monkey brains over an extended period of time1. After removing a patch of skull, researchers insert a thin, circular piece of glass ringed with a titanium wall — all of which is surrounded by a flexible polymer membrane.
The titanium wall and membrane prevent a problem that often plagues these experiments in monkeys: the obscuring of the glass window after the membrane covering the brain regenerates.
The researchers also developed an algorithm that counteracts image distortion from movement arising from breathing or pulse — another common issue.
When the researchers implanted the window in a monkey’s skull, the images they created were sharp enough to resolve the delicate branches of neurons, called dendrites. The novel window and algorithm allowed the researchers to monitor firing neurons in four monkeys up to five months after surgery, and the window remained clear even at nine months.
Deep dive:
Another technique uses long wavelengths of light to illuminate neuron activity deep in the mouse brain2. An approach called two-photon microscopy can resolve this activity in the outer layers of the brain. But deeper neurons are harder to visualize because tissue scatters and absorbs light.
The new technique, called three-photon microscopy, reveals neurons firing in the mouse hippocampus, an embedded brain region involved in memory.
The researchers used a custom-built microscope to record the activity of 150 neurons at once. The images are about 40 times sharper than those of the hippocampus captured using two-photon microscopy. The method appeared 20 February in Nature Methods.
Precise probe:
An alternative way to see the brain’s interior is to implant fiberoptic probes that deliver light directly to the neurons of interest. But these probes are thick and can damage the surrounding tissue.
To minimize damage, researchers used an ultra-thin glass needle to deliver light 2 millimeters into a dissected mouse brain. The needle’s diameter is one-fifth the size of conventional fiberoptic probes.
The technique caused minimal tissue damage and allowed researchers to distinguish fine cellular features, such as the branching protrusions of brain immune cells called microglia, according to a study published 20 March in Scientific Reports3.
With slight modifications, the researchers say the system could work in awake, moving mice.
Quick scan:
A standard two-photon microscope scans brain tissue plane by plane and reconstructs the snapshots into a 3-D image. This process is too slow to capture large neural networks in action. But a tweak to the setup, described 27 February in Nature Neuroscience, allows researchers to scan through the sample quickly4.
The tweak involves reshaping the laser light from a two-photon microscope into a ring-like beam that the tissue does not diffuse.
When aimed at neural tissue in fruit flies, zebrafish larvae, mice and ferrets, the ring-shaped beam covers an area that requires 36 frames to capture the standard way. As a result, the researchers were able to create high-resolution videos of neurons firing across large swaths of brain tissue.
By joining the discussion, you agree to our privacy policy.