Matt Judson and Alain Burette, UNC
THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
Editor’s Note
Some of the findings discussed in the conference report below were published 29 June 2017 in the Journal of Neuroscience1.
Unusually thin nerve fibers in the brain may underlie the motor difficulties seen in some children with Angelman syndrome, an autism-related condition. The unpublished findings were presented Thursday at a conference in Silver Spring, Maryland, hosted by the Dup15q Alliance and the Angelman Syndrome Foundation.
The findings point to a possible new biomarker for the condition, and even a road toward a treatment.
People with Angelman syndrome tend to have difficulty with balance, walking and manipulating objects with their hands. Other features include a small brain, happy demeanor, intellectual disability, seizures and lack of speech.
Angelman syndrome typically results from the deletion of a large region of chromosome 15 called 15q11-13. But mutations in UBE3A, a gene in the region, are sufficient to cause the syndrome. Having an extra copy of UBE3A or of 15q11-13 is a strong risk factor for autism.
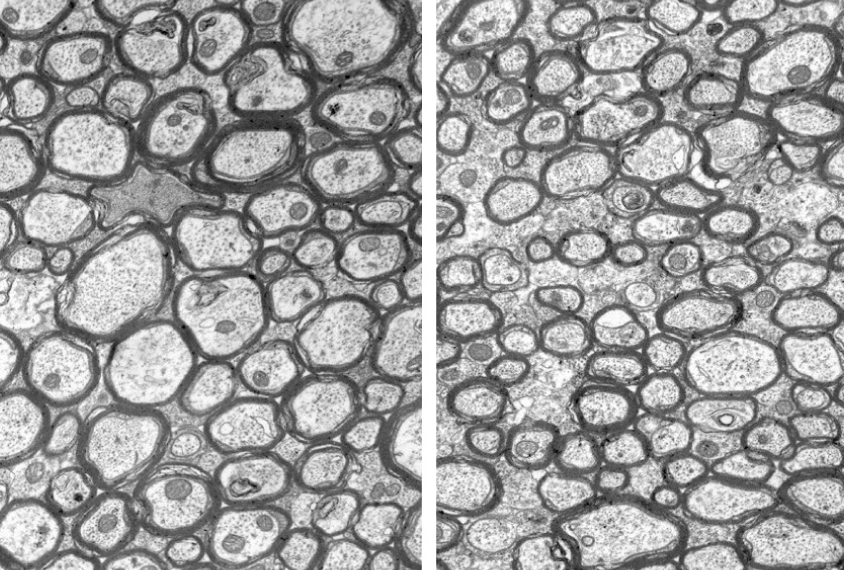
The new study shows that children with Angelman syndrome have alterations in white matter, the fibers that extend from neurons. The researchers also found that the brains of mice lacking UBE3A expression in neurons mirror these alterations, which they traced to unusually thin nerve fibers. Partially restoring UBE3A expression in some mouse neurons enlarges the brain slightly, but the researchers do not know why this happens.
The findings hint that normalizing UBE3A expression in people with Angelman syndrome might repair a key structural problem in their brains. “If we can add the gene back early enough, then we might be able to reverse the white matter deficits or prevent them from continuing to emerge,” says Benjamin Philpot, associate director of the Neuroscience Center at the University of North Carolina at Chapel Hill, who presented the findings.
“This work is really important because it shows, at the brain level, potential mechanisms for why these kids have delays,” says Shafali Jeste, associate professor of psychiatry and neurology at the University of California, Los Angeles, who was not involved in the work. If the findings in mice apply to people, she says, white matter structure might serve as a biomarker for monitoring the effectiveness of Angelman treatments.
Imaging integrity:
Philpot’s team used magnetic resonance imaging (MRI) to scan the brains of 11 children with Angelman syndrome and 40 controls, all 3 to 9 years old. The scans revealed that the children with Angelman syndrome have unusually small brains sporting 30 percent less white matter than typical children. Their brains also have 16 percent less gray matter, which contains the cell bodies of neurons.
The less white matter a child with Angelman syndrome has, the more severe his or her motor difficulties, as measured by caregiver reports and lab tests.
The researchers then used a method called diffusion tensor imaging to probe the structural integrity of two nerve fiber bundles involved in motor function. These are the corpus callosum, which bridges the brain’s hemispheres, and the corticospinal tract, which connects the brain and spinal cord.
These tracts have less structural integrity in children with Angelman syndrome than in typical children. And the greater the structural alterations, the more severe a child’s movement difficulties, Philpot reported.
“Their motor tracts are not as well organized,” says Heather Hazlett, assistant professor of psychiatry at the University of North Carolina at Chapel Hill, who led the brain imaging work. She says that the team is looking to see whether the same anatomical oddities appear in children with other neurodevelopmental conditions, including autism.
Mouse mimic:
The researchers used MRI to examine white matter volume and structure in mice missing a copy of UBE3A. They found that the mice have alterations that parallel those seen in the children with Angelman syndrome.
In brain tissue from the mutant mice, the researchers found a normal number of nerve fibers in the corpus callosum. The fibers have typical amounts of myelin, a waxy substance that insulates them, and they extend to the right places in the brain. But they have one curious feature: They are unusually thin.
“We’ve really narrowed down the defect to a selective reduction in diameter,” Philpot says.
The researchers bred the mutant mice so that their pups regain normal UBE3A expression levels in a subset of neurons. These pups developed slightly larger brains than did those lacking UBE3A expression throughout the brain.
“Very early UBE3A reinstatement might alter the trajectory of white matter development,” Philpot says.
If the findings hold up in people, monitoring white matter changes may enable researchers to track treatment efficacy. “We’re really on the cusp of having a slew of therapeutics coming into clinical trials, and successful clinical trials will largely depend on having good biomarkers,” Philpot says.
Hurdles remain, however. “To use it as a biomarker for response to a treatment, you would have to know how quickly it changes,” says Sarah Spence, assistant professor of neurology at Harvard Medical School, who was not involved in the work. If white matter changes at all, the process may take longer than the typical run of a clinical trial.
In the meantime, looking at white matter in a child with Angelman syndrome could help doctors provide a prognosis for the timing and severity of any motor problems that might develop.
For more reports from the 2016 Dup15q Alliance Scientific Meeting, please click here.
By joining the discussion, you agree to our privacy policy.