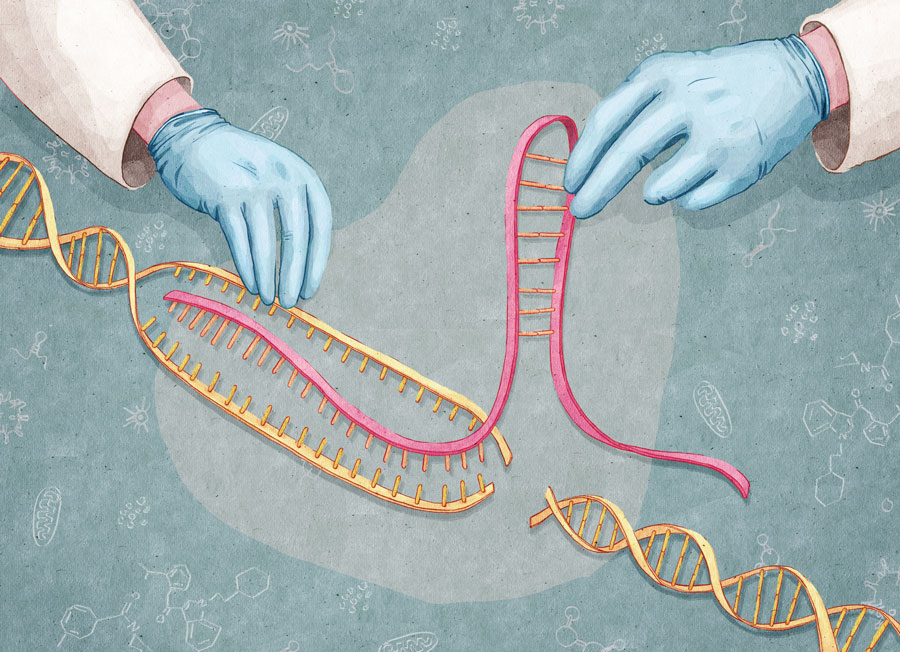
Gene-editing tool hews shortcut to autism mouse models
A widely used gene-editing technique can create large chromosomal defects in mice, slashing the time and effort needed to engineer mouse models of these genetic glitches.
A widely used gene-editing technique can create large chromosomal defects in mice, slashing the time and effort needed to engineer mouse models of these genetic glitches. The advance was described 4 February in Cell Reports1.
CRISPR is commonly used to add, delete or change sequences within genes. Researchers are using it to make human cells carrying mutations for Angelman syndrome and Rett syndrome, two autism-linked disorders.
In the new study, researchers modified CRISPR to generate large duplications and deletions of DNA, called copy number variations, in mouse embryonic stem cells. They also used the method to create inversions — chunks of DNA that have been cut out of the genome and reinserted in the opposite direction. Then they used the modified stem cells to create mice carrying the variants.
“Our method enables researchers to create such structural variations in mice in a very short time,” says lead researcher Stefan Mundlos, head of the development and disease group at the Max Planck Institute for Molecular Genetics in Berlin, Germany. “It allows us to create any kind of change in order to investigate how the structure of the genome influences gene regulation, for example.”
Scientists have traditionally used a long and tedious process called chromosome engineering to create mice carrying large chromosomal mutations. The process involves three lengthy steps: two to mark the ‘cut’ sites at each end of the sequence and a third to cut it out and delete, duplicate or invert it. Once researchers isolate the cells with the alteration, they can use the cells to create a colony of mice.
Timesaving tool:
The new approach, dubbed CRISVar, allows researchers to make the cuts and the chromosomal alteration all at once. “You can basically generate everything in the same experiment,” says Darío Lupiáñez, a postdoctoral researcher in Mundlos’ lab. Lupiáñez and Guillaume Andrey, another postdoc in the lab, developed the technique together.
The technique gives researchers a quicker and easier method than chromosome engineering to study autism-linked chromosomal variations, says Alea Mills, professor at Cold Spring Harbor Laboratory in New York, who was not involved in the study.
Mills helped to develop chromosome engineering in the 1990s, and used it in 2010 to generate mice that model a deletion or duplication in the 16p11.2 chromosomal region, risk factors for autism.
“This new procedure is very simple, and pretty much anybody can use it.”
“People are terrified to use the type of technology that we’ve been using because it takes a long time and a lot of expertise,” Mills says. “But this new procedure is very simple, and pretty much anybody can use it.”
CRISPR, which can be used to edit virtually any sequence in the genome, debuted two years ago2,3. Researchers simply supply cells with the DNA-snipping enzyme CAS9 and a guide RNA that steers the enzyme toward the desired genetic region.
In the new study, researchers infused cells with two guide RNA molecules — one that pilots CAS9 to the beginning of the region and a second that guides it to the end. The enzyme then snips through both DNA strands at each site, freeing the intervening sequence.
In response, the cell tries to stitch the fragmented ends back together. The process is imprecise, however, and can patch the region in various ways — deleting, duplicating or inverting the targeted region in the process.
The researchers applied the technique at six sites in the mouse genome, ranging in size from 1,000 to 1.6 million base pairs. They were able to generate deletions or inversions at each site and duplications at four of them. Intriguingly, the size of the target region seems to have little influence on the method’s efficiency.
One of the target regions encompasses 353,000 base pairs of the LAF4 gene. Deletion of this region mimics a deletion found in a person with limb malformations. The researchers generated mice from cells with the deletion in one or both copies of the gene, and found that the animals show similar limb abnormalities.
Precision problem:
The entire process — from design of the guide RNAs to the mice — took only 10 weeks, compared with the 3 to 12months it can take just to alter mouse embryonic stem cells using chromosome engineering.
“According to their results, just about any deletion or inversion can be created,” says Jonathan Sebat, chief of the Beyster Center for Molecular Genomics of Neuropsychiatric Diseases at the University of California, San Diego. Sebat was not involved in the work, but has shown that CNVs occur more frequently than usual in people with autism. “This is very encouraging if it works efficiently at most sites.”
Sebat and others caution that the approach needs to be tested in a wide array of sites to confirm that it will work anywhere in the genome. Some regions may be less accessible to cutting and therefore more difficult to rearrange than others, for example.
The researchers say the method’s efficiency varied even among the six sites they tested, for reasons they don’t yet understand. For instance, they were able to induce deletions in 42 percent of cells at one site, but just 2 percent at another. “We don’t really have an explanation of how all this works molecularly,” says Andrey.
In some instances, the researchers found a mix of chromosomal abnormalities in the same cell. “You might want to generate a deletion, but you also need to carefully check for everything [else] that could be happening there,” Lupiáñez says. “You really don’t know what the cell is doing there.”
The approach may also alter regions other than the intended sites, says Nicholas Katsanis, director of the Center for Human Disease Modeling at Duke University in Durham, North Carolina. Katsanis was not involved in the new study, but has used CRISPR extensively in zebrafish and is generating mouse models of autism-linked CNVs.
“No matter what, it’s going to be more efficient than traditional tools,” Katsanis says. “But let’s not imagine a world where you just flick a switch and you have your deletion made.”
References:
1. Kraft K. et al. Cell Rep. Epub ahead of print (2015) PubMed
2. Cong L. et al. Science 339, 819-823 (2013) PubMed
3. Mali P. et al. Science 339, 823-826 (2013) PubMed
Explore more from The Transmitter
Cocaine, morphine commandeer neurons normally activated by food, water in mice
X chromosome inactivation; motor difficulties in 16p11.2 duplication and deletion; oligodendroglia
