THIS ARTICLE IS MORE THAN FIVE YEARS OLD
This article is more than five years old. Autism research — and science in general — is constantly evolving, so older articles may contain information or theories that have been reevaluated since their original publication date.
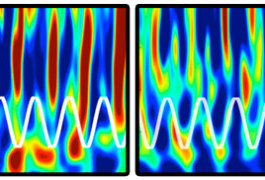
PAC a punch: The coupling of alpha waves (white) and gamma bursts (top, red) is different in individuals with autism (right) than in controls (left).
Connections between neighboring groups of brain cells are weaker in individuals with autism than in controls, according to a report published 14 January in the Proceedings of the National Academy of Sciences1.
This is the latest of several studies in the past year to cast doubt on the so-called ‘connectivity theory’ of autism, which broadly says that people with autism have stronger connections between neighboring brain regions and weaker connections between distant regions than controls do.
Using magnetoencephalography (MEG), a noninvasive imaging technique that uses magnetic fields to track rapid changes in brain waves, the new study found that both short- and long-range connections are impaired in autism.
What’s more, the weakest local connections are found in those with the most severe autism symptoms, the study found.
That both short- and long-range connections are weaker in autism makes intuitive sense, says lead investigator Tal Kenet, an instructor in neurology at Harvard Medical School.
“What always bothered me about the [connectivity] hypothesis was, why would neurons be really bad at long-range but good at it locally?” she says.
The new findings support a more general hypothesis — supported by genetic data — that autism is a broad failure of communication among neurons, she says.
The connectivity hypothesis first became popular about a decade ago. In a 2004 report, researchers found that when individuals with autism perform a sentence comprehension task, distant regions of the brain are not as synchronized as they are in controls2.
Since then, many studies have bolstered the idea of weak long-range connectivity in autism. For example, in September, researchers reported that children carrying the autism-linked MET gene have weak long-range connections in the cortex, or outer regions of the brain.
Local source:
Research on local connectivity, however, is scant.
The first evidence for excessive short-range connections dates to postmortem studies in 20023. Studying these connections in living people is difficult, however, because of the technical limitations of brain scanners.
The commonly used functional magnetic resonance imaging (fMRI) uses blood flow as a proxy for neuronal activity. Local connectivity is difficult to track with this method because neighboring brain areas depend on the same blood supply, according to Kenet.
“If you try to look at two areas that are very nearby each other, you’re always going to see very, very high coordination, just because of the artifact of the method,” Kenet says.
Special Report: Connectivity
This is one of a series of articles exploring brain connectivity in autism.
Read more »
Her team instead used an indirect marker of local connectivity.
The firing patterns of large groups of neurons produce different kinds of brain waves, some fast and some slow. Studies of people getting brain surgery for epilepsy have revealed that repeated slow waves, called alpha, can trigger bursts of fast waves, called gamma.
This particular pairing — called phase-amplitude coupling, or PAC — comes from small (on the order of a centimeter) areas in the brain4.
These brain-wave patterns happen in a matter of milliseconds — far too quickly for fMRI, which tracks activity from second to second. MEG, in contrast, has much better temporal resolution.
The new study used MEG to measure PAC in 17 young men with autism and 20 controls as they looked at pictures of houses, neutral faces, angry faces and fearful faces.
The researchers looked at local connectivity in a 0.65 square-centimeter area of the fusiform face area (FFA), a region in the cortex that is active during face processing.
People with autism show weaker PAC — that is, less synchrony between alpha and gamma waves — when looking at faces than controls do, the study found. They also have weaker long-range connections between the FFA and other areas of the cortex than controls do.
“I think this is probably one of the strongest electrophysiology studies looking at connectivity in autism [up] to this point,” says Ralph-Axel Müller, professor of psychology at San Diego State University in California, who was not involved in the work.
Connectivity confusion:
The findings are consistent with a 2011 study by Müller’s team using a different technique, called diffusion tensor imaging. In that study, the researchers reported that bundles of white matter spanning short distances are compromised in children with autism5.
However, it’s difficult to compare connectivity findings from one study to the next, Müller says. For example, the epilepsy studies showed that inhibitory neurons drive PAC signals, whereas fMRI — used in most connectivity studies of autism — measures excitatory signals.
If a weaker PAC means fewer inhibitory connections, the data could go along with a long-held theory that autism stems from an imbalance of excitatory and inhibitory signals. “The differences in results would then be expected rather than surprising,” Müller says.
It’s also unclear how the new results fit in with the original postmortem studies. In that work, Manuel Casanova and his colleagues reported that postmortem cortical tissue from individuals with autism shows abnormally narrow and dense stacks of excitatory neurons and fewer inhibitory neurons than in tissue from controls.
“It’s like apples and oranges,” says Casanova, professor of psychiatry at the University of Louisville. “Histology and anatomy have very different implications [than do] physiology and function.”
Kenet says she doesn’t think her new findings contradict the postmortem work.
“I think it’s actually completely in agreement,” she says. If a baby’s brain has too few inhibitory neurons, it would probably compensate for that during development, perhaps resulting in weaker short- and long-range connectivity, she says. “The brain is a very dynamic entity.”
If confirmed as specific to autism, PAC may one day be used as a diagnostic biomarker of autism, the researchers say. It may be particularly useful for detecting the disorder in younger siblings of children with autism, who have a higher risk of developing it compared with the general population.
References:
1. Khan S. et al. Proc. Natl. Acad. Sci. U.S.A. Epub ahead of print (2013) PubMed
2. Just M.A. et al. Brain 127, 1811-1821 (2004) PubMed
3. Casanova M.F. et al. Neurology 58, 428-432 (2002) PubMed
4. Canolty R.T. et al. Science 313, 1626-1628 (2006) PubMed
5. Shukla D.K. et al. Neuropsychologia 49, 1378-1382 (2011) PubMed
By joining the discussion, you agree to our privacy policy.